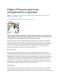
Pharmacy
Content
Pharmacy Sr. No.
Core Areas
Percentage
1.
Pharmaceutics
20%
2.
Pharmaceutical Chemistry
20%
3. 4.
Pharmacology Pharmacognosy
20% 20%
5.
Practice of Pharmacy
20% Total
100%
Pharmacy (Detailed) Sr. No.
Core Areas
Percentage
PHARMACEUTICS: 1. Pharmaceutical Principles and Drug Dosage Forms______6% 1.1 Physicochemical Behaviors: 1.1.1 Homogenous Systems: Solutions: Solutions: Solvents, Solutes, Electrolytes, Non-electrolytes 1.
Colligative properties: properties: Lowering of vapor pressure, Elevation of boiling point, Depression of freezing point, Osmosis and Osmotic pressure. Buffers and buffer capacity: What are the buffers, Buffer action, and Buffer capacity? 1.1.2 Heterogeneous (Disperse) Systems: Basics about Suspensions and Emulsions Dispersion stability: stability: Ideal dispersion, Particle size, Dispersed phase concentrations, Particle-particle interactions, Density.
Emulsion Stability: Stability: Creaming, Coalescence, electrostatic repulsion, Phase inversion, Cracking. 1.2 Chemical Kinetics and Drug Stability: Introduction to stability of active ingredients: Reaction rates and orders of reaction. Factors affecting the reaction rates: Temperature, Presence of Solvent, Change in pH, Presence of additives. Modes of pharmaceutical Degradation: Hydrolysis, Oxidation, Photolysis. 2. Drug Dosage Forms ________________________ ______________ _____________________6% ___________6%
20 %
Basics of Dosage forms: Tablets: Definition, Types, Advantages and disadvantages, Methods of Manufacturing, Excepients, Tablet defects, Coating effects. Capsules: Definition, Types, Methods of Manufacturing, Excepients. Oral drug solutions: solutions: Syrups and their additives, elixirs and their additives. Suspensions: Types, Purpose of suspensions, Suspending agents. Emulsions: Emulsions: Types, Purpose of emulsion, Phases of emulsions, Emulsifying agents, Creams and Ointments; Introduction, Types of creams and ointments, Types of their bases, methods for preparation. Suppositories: Introduction, Types, Suppository bases, methods for preparation. Powders: Powders: Definition, Advantages and Disadvantages, Micromeretics, Trituration, Pulverization, Levigation, Spatulation, Simple and geometric mixing, Hygroscopic, Deliquescent substances, Eutectic mixtures. Aerosol Products: Products: Introduction, Advantages and disadvantages, Aerosol additives. Sterile Products: Products: Sterility, Introduction, advantages and disadvantages of additives of Injectable. 3. Biopharmaceutics and Drug Delivery Systems __________8 % Definitions: Definitions: Biopharmaceutics, Drug Product, Pharmacokinetics, Pharmacodynamics, Bioavailability, Bioequivalence, Absorption, Distribution, Elimination.
Biopharmaceutical Principles: Principles: Drug Dissolution, Drug Solubility, Particle size and surface area, Partition coefficient and extent of ionization, Salt formation, Polymorphism, Chirality, Hydrates, Complex formation. Basic Pharmacokinetics: Zero order and first order reactions, Models and compartments, Drug Distribution and elimination. Bioavailability Bioavailabili ty and Bioequivalence: Bioequivalence: Relative and Absolute bioavailability, AUC, Cmax, Tmax, Half-life, Sustained Release/Controlled Release Dosage forms, Introduction, Microencapsulation, Microspheres/Microcapsules, Microspheres/Microcapsu les, Dose dumping. PHARMACEUTICAL CHEMISTRY: 1. Instrumentation____________________ Instrumentation________________________________ ___________________6 _______6 % Basic Principle, instrumentation and Pharmaceutical
2. 2.
applications of following techniques; HPLC, UVspectrophotometery, IR 2. Chemistry of Biomolecules_____________________ Biomolecules_____________________________7% ________7% Introduction, Biological and pharmaceutical importance of Carbohydrates, Proteins, Lipids, Vitamins, Enzymes and Hormones 3. Principles of Medicinal Chemistry ______________________ ______________________7% 7%
20 % %
3.1 Structure Activity Relationship; 3.1.1 Structurally non-specific drugs, 3.1.2 Structurally specific drugs Receptor-site theory, Receptor-site Binding, Stereochemistry (optical isomers, geometric isomers and conformational isomers) PHARMACOLOGY: 1. General Pharmacology__________________ Pharmacology_______________________________ _____________ 7 %
3. 3.
Introduction,Pharmacokinetics,Pharmacodynamics, Dose, Efficacy, Safety, Potency, Dosage, Drug-Dose Response curve, Therapeutic index, Receptors, Agonists and antagonists, Pharmacological antagonism (Competitive and Non-competitive), Partial antagonist, Physiological antagonism, Neutralizing antagonism. 2. Mechanism of Drug Actions _______________________ ___________________________7 ____7 %
20 % %
Cell surface receptors, Signal transduction by cellsurface receptors, Signaling mediated by intracellular receptors, Target cell desensitization and hypersensitization, hypersensitizat ion, Pharmacological effects not mediated by receptors (for example anesthetics and cathartics). 3. Pharmacology of commonly used drug classes________6 % Classification, mechanism of action, indications and adverse effects of following commonly used drug classes; analgesics/antipyretics, anti-infective, antihypertensive, Diuretics, Anti-parasitic, Anti-malarial, drug sued in gastrointestinal gastrointestinal disorders. PHARMACOGNOSY: 1. General introduction ________________________________ __________________________________7 __7 % Classification of Crude Drugs with Special Emphasis to Chemical and Therapeutical System of Classification.
4. 4.
Terminologies used in Pharmacognosy; Pharmacognosy, crude drugs, Preparation of Crude Drugs for Commercial Market, Methods of Cultivation, Drying, Storage. Preservation, Packing, Deterioration and Adulteration of Crude Drugs. Evaluation of Crude Drugs (Organoleptic, Microscopic, Physical, Chemical and Biological).
20 % %
Allergens and allergenic preparation_____________________7 preparation_____________________7 % Introduction, case history, skin test, treatment off allergy, inhalant, ingestant, injectant, contactant, infectant and infestant allergens. Mechanism of allergy. 3. Separation and isolation of plant constituents___________6 constituents___________6 % An introduction to chromatography and chromatographic techniques e.g. Adsorption Chromatography and Partition Chromatography. Chromatography. PHARMACY PRACTICE: 1. Community/Retail Comm unity/Retail Pharmacy __________________________ __________________________4% 4% 5. 5.
Basic introduction about; Prescription (parts, handling and filling of prescriptions), Medication order, Difference between Prescription and Medication order, Dose calculations, Compounding, Extemporaneous
20 % %
preparations, Pharmaceutical Care, Dispensing, Epidemiology, Drug abuse & Misuse, Patient Medication Record. 2. Medication errors_______________________ errors__________________________________ _______________4% ____4% Definition, Types of errors; Wrong drug error, Extra dose error, Omission error, wrong strength error, Wrong route error, Wrong time error, Wrong dosage form errors. 3. Clinical Pharmacy _______________________ ____________________________________ _____________4% 4% Clinical Pharmacokinetics, Therapeutic Drug Monitoring (TDM), Clinical trials, Importance of Clinical Pharmacy, Pharmaco-economics, Pharmaco-vigilance, Patient Assessment, Patient Counseling, Patient Compliance/non-compliance, Compliance/non-complian ce, Rational use of drugs, Drug Utilization Evaluation, Drug Utilization Review. 4. Adverse Drug Reactions__________________ Reactions_______________________________ _____________4% 4% Definition and difference between Adverse drug reactions and side effects, Types of Adverse drug reactions (Type-A and type-B). 5. Drug interactions____________________ interactions_________________________________ ________________4% ___4% Mechanism, Physiological factors affecting interaction, Types and level of drug interactions. Total
100%